
The temperature at which a substance condenses is the same as the temperature at which it boils. Substances undergo condensation when they change from a gas to a liquid.Ĭondensation can be observed on a cold glass of water on a hot summer day. Cooling a substance enough can cause a change in its state. When heat is removed from a substance the particles lose energy, slow their motion, and pack together more tightly. When liquids reach a certain temperature their particles gain enough energy to escape the surface of the liquid and change into a gas. Particles in a liquid continue to move even faster and spread further apart when more heat is added. When enough heat is added to a solid it can change into a liquid. When heat is added to a solid the particles gain energy and they move around faster and spread further apart. Frost can also wipe out entire crops if an unexpected freeze happens in the late spring. Another negative aspect is that when pipes freeze in the winter they can burst. On the other hand, water in cracks on roads freezes in the winter and expands, as this happens over and over, small cracks can become large potholes. If ice sank than the entire lake would freeze each winter. When lakes freeze the ice is able to insulate the water below so it does not freeze and fish do not die. 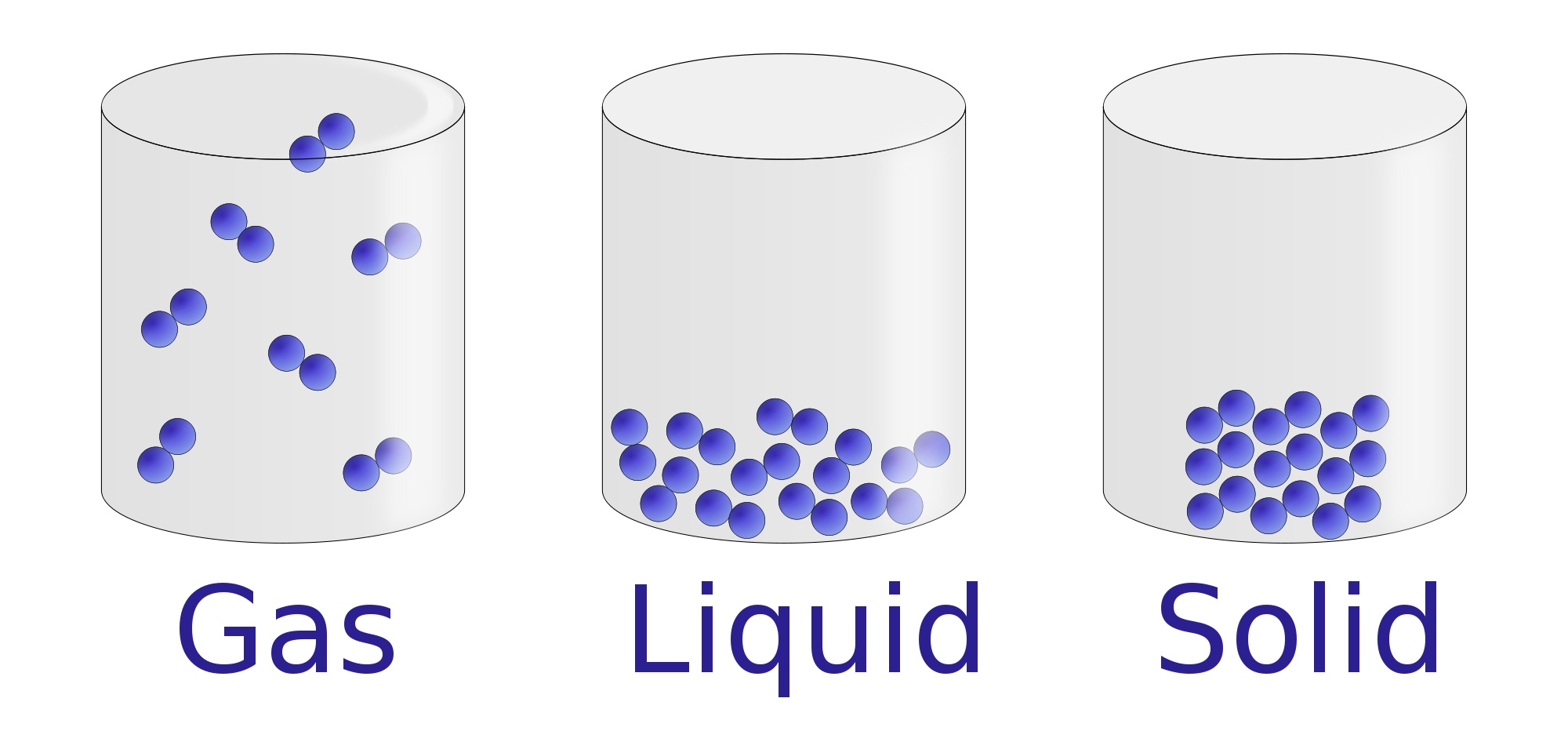 
There are some positive and negative implications of the fact that ice is less dense than water. Ice floats on top in a glass of ice water. Ice is actually less dense then liquid water. When heat is removed from water it freezes becoming ice. One exception to this rule of heat affecting density is water. When heat is removed the opposite happens and particles slow down and move closer together, therefore becoming denser. After adding even more heat, the particles spread farther apart and expand, thus becoming a gas, which is much less dense. This can overcome the bonds holding them together causing a solid to become a liquid. When heat is added particles move faster. Substances change their state of matter when heat is added or removed. 
Liquids and gases are known as fluids because both substances are able to flow. Since gases do not have a definite volume they can spread out in all directions. Gases do not have definite shapes or definite volumes and like liquids gases take the shape of their containers. Unlike solids, liquids take on the shape of their container. Solids are substances that have both definite shapes and definite volumes.Ī liquid, on the other hand, has a definite volume, but does not have a definite shape. Gases therefore do not have a definite shape or definite volume.įigure 1. Particles in a gas are able to move around freely and it is this movement that allows gases to expand or contract. Gases are the state of mater that have the weakest bonds between their particles. Since the particles can move, liquids are able to change their shape and conform to the container that holds them. The bonds that hold the particles of a liquid together are weaker then in solids, which is the reason that particles in a liquid slide and move past one another. Liquids have particles that are able to move throughout the container that the liquid is in. It is these strong bonds that give solids their definite shape and volume. Particles in a solid, as a result, vibrate in their positions. The energy that bonds these particles together is very strong and prevents the particles from moving around freely. In a solid the particles are packed tightly together. The motion and arrangement of the particles that make up a substance determines its state of matter. These particles have energy and are in a constant state of motion. Matter is made up of very tiny particles known as atoms and molecules.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
